
Sleep is probably one of the most fascinating states of the mind and body. A good night’s sleep is essential for a fully functioning day ahead, so essential in fact, that this incredibly natural process has had years and years of research devoted to it. Numerous studies are conducted worldwide to further explain everything from why we sleep, to the numerous factors behind how we maintain prolonged sleep, to sleep quality. But in this fray, there is one area that is understudied – sex differences in sleep. Do women sleep different than men? Is their quality of sleep different? Or perhaps the hours they spend in the state? Work in the Einstein Lab aims to understand some of these questions, starting with estrogens – the hormones with so many roles and functions, many still not properly understood.
Dr. Nicole Gervais, a sleep researcher in the Einstein Lab, has been studying the impact of ovarian hormones on memory and sleep for several years now. “My focus started in animals including rats and marmosets, and I’ve now moved up to women,” she says. “Now I study menopausal women mostly, and the focus for me is understanding how hormone-related changes in sleep can increase women’s risk for Alzheimer’s disease.”
Sleep and Estrogen in Female Marmosets
Dr. Gervais first started getting interested in sleep during her first post-doctoral fellowship at the University of Massachusetts Amherst. “We did a study on ovariectomized marmosets to try to see whether we can model some of the menopausal symptoms that women report,” she recalls.
Marmosets are New World Monkeys native to Brazil. They have white blazes on their foreheads and white ear tufts, and are tender, intelligent animals. According to Dr. Gervais, her former lab at UMass studied sleep in marmosets because they’re diurnal primates (their sleep cycles matches that of humans) and their relatively short lifespan makes them a good model for aging. However, they do not experience natural menopause, and so to study the influence of ovarian hormones on sleep their ovaries are removed (which is more of a model of surgical menopause in humans.)

“We looked at cognition and sleep [in these marmosets] and modelled hot flashes,” Dr. Gervais details. “And then we wanted to see what impact estradiol [most comment and potent form of estrogen] would have on the symptoms. Basically, the focus ended up being on sleep as we found really interesting effects of applying estradiol and then taking it away,” she says. “We saw that under low estradiol levels the marmoset’s sleep improved and when you took the estradiol away, they started showing sleep changes.” Dr. Gervais also mentions that she and her team saw corresponding effects of temperature, particularly at night. “So under estradiol core temperature during sleep withdrew more so it made them less likely for them to be able to get hot flashes, and lower temperature is also better for sleep overall,” she explains.
After her work on marmosets, Dr. Gervais’ interest was piqued on the possible relationship between estradiol and sleep in women. In the Einstein Lab, she is investigating this relationship in those with a bilateral salpingo-oophorectomy, who carry the breast cancer mutations BRCA1 and BRCA2 and have chosen to remove both their ovaries and fallopian tubes in an effort to allay the risk of breast or ovarian cancer. In addition to higher risk of Alzheimer’s disease, these women also have higher risk for sleep disorders, including insomnia and sleep apnea. These disorders themselves place women at higher risk for AD. These women with bilateral salpingo-oophorectomies help researchers such as Dr. Gervais investigate the impact of estrogen loss on cognition in women after menopause – in which women undergo a similar decrease of estrogen – but without the confounding effects of age.
How is Sleep Measured?
“When women come into the lab, we teach them how to wear a polysomnography device,” Dr. Gervais says. “[It contains] a recorder that sits on a belt that goes on the chest. A corresponding one goes on the abdomen. Both of these are going to record your breathing while you sleep.” The polysomnography device that she and her team use contains a headband with five electrodes that are attached on various points on the face (including 2 that measure brain activity using what is called electroencephalography). The women take home the device and wear it for three nights, giving Dr. Gervais’ team ample data to analyze. “In addition to the electrodes and the chest bands we also have an oximeter, which measures pulse and oxygen desaturation (via SpO2), which now is a common term everyone knows because of the coronavirus,” she says. Measuring the pulse and SpO2 – the amount of red blood cells carrying oxygen relative to the amount not carrying oxygen – help Dr. Gervais assess sleep apnea, a sleep disorder in which breathing repeatedly stops and starts and is common in post-menopausal women. It is also a risk factor for developing Alzheimer’s Disease.
Dr. Gervais and her team then analyze the brain activity data from the polysomnography device, which yields sinusoidal waveforms, data that is captured throughout the night while the participant sleeps. This data tells them which stage of sleep the participant is in (known as sleep architecture) and provides information on the quality of sleep. “And we can look at how [oophorectomy] can change those waveforms in women,” she says.
What did they find?
“We’re still analyzing data, but so far what we’re seeing is that women with BSO [bilateral salpingo-oophrectomy] are reporting subjectively poor sleep quality, but we’re also seeing from the polysomnography device they’re taking longer to fall asleep,” Dr. Gervais reports. According to her, these symptoms are consistent with those of insomnia. “They’re also experiencing more cortical arousal. [During this period] a very short burst of higher frequency of activity while they’re sleeping occurs, which is indicative of less restful sleep,” she says. This information is indicative that removing the ovaries of a woman has a drastic effect on her sleep, and ovarian hormones are primarily responsible for this. “I can’t say it’s estradiol alone because some of the women who are taking in our study are taking progesterone [another important ovarian hormone],” she reasons. “In addition, and we know progesterone has actually a hypnotic effect, and so it will improve your ability to fall asleep but it doesn’t necessarily lead to better sleep quality.” Dr. Gervais believes that this decline in ovarian hormones is why women experience poor sleep after menopause, since their bodies have stopped producing them.
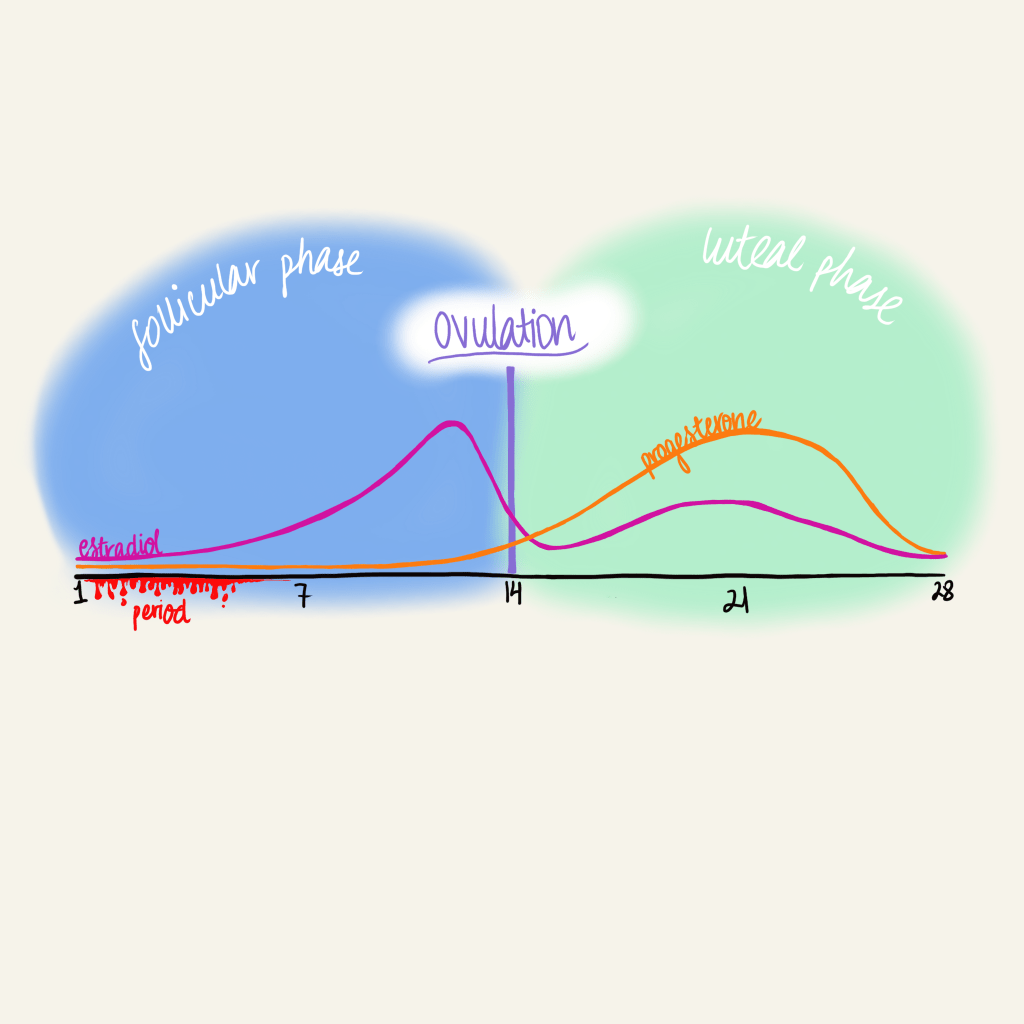
This data is not only informative for women undergoing menopause and with oophorectomies, but pre-menopausal, menstruating women as well. “Just to recap, our menstrual cycle is broken up into two phases – the follicular phase and the luteal phase – and the beginning part of the early follicular phase is when [we] undergo [menstruation],” Dr. Gervais explains. “And then during the late follicular phase is when we ovulate, after which we switch over to the luteal phase. So it’s during this late follicular [phase] that we have a huge increase in estradiol, and then during the luteal phase we see a moderate level of estradiol but also progesterone, and the both hormones start to decline (at the end of the luteal phase).” Dr. Gervais recounts findings from others showing that the late luteal phase – when both estradiol and progesterone are declining – is associated with worse sleep. “The luteal phase is also associated with a decline in something called sleep spindles, events [during sleep] which include a sharp burst of [brain] activity, that’s important for memory consolidation,” she explains. “We don’t know the significance of this but that’s very interesting.”
Estrogen and Dreams
When asked about any possible consequences of ovarian hormone levels on dreams, Dr. Gervais replied, “Let’s just talk about it in the context of REM sleep because that’s when we think we’re dreaming. According to Dr. Gervais, REM sleep is important for our moods, as if we don’t get enough REM sleep the night before, our mood the next day suffers. “So we do know that estradiol loss at least in the animal models can alter REM sleep in terms of duration,” she says. “We haven’t necessarily seen that observed in in the humans so much. One of the things that this research area is struggling with is being able study in the rodent and observe the same kind of findings in humans. There’s a lot of challenges there. Also, this is overall an understudied area, but it’s a really interesting question that what would estradiol do to our dreams. We’re not seeing right now any changes in REM sleep as a function of estradiol in humans as far as I’m aware, but what impact it can have on dreams, who knows. That’s a really great question maybe neuro imaging studies can help us understand that.”
Currently, the COVID-19 pandemic has put efforts for this project on pause. But when operations are running again, Dr. Gervais hopes to make some real strides on this project. “We’re going to continue collecting the data, and we have two more PhD students on board,” she says. [One of them], Laura Gravelsins, is interested in the influence of different genes on sleep, and [another], Alana Brown, is interested in how sleep can impact associative memory.” The Einstein Lab has also pioneered a new online study during this period, which includes questions on insomnia. “Right now, I’m interested in looking at the importance of insomnia in menopausal women, [women with oophorectomy] in in particular,” Dr. Gervais says. “Insomnia is a risk factor for Alzheimer’s disease, much like sleep apnea which is also a risk factor, but I’m wondering if insomnia might be a female specific risk factor for Alzheimer’s Disease. Women are more likely to get insomnia than men and their prevalence increases after menopause. And if you don’t treat insomnia, and if someone’s left with this condition for a long time, this will have permanent effects on the brain we think. And we don’t really know what impacts it has, since people haven’t studied the long-term effects of insomnia following menopause and dementia risk. So that’s one of the questions I have,” she explains.
Ultimately, Dr. Gervais and her team hope to make some relevant contributions to the understudied field of sleep and women and hope to lay some groundwork for more research on the subject. Their work is supported by Brain Canada and the Alzheimer’s Association.